An overview of Emergency Use Authorization and how it differs from normal vaccine approval.
The U.S. Food and Drug Administration (FDA) has released the first three COVID-19 vaccines for emergency use and others are expected to follow. Some consumers may have questions about what Emergency Use Authorization means, and how it differs from normal vaccine approval.
In a public health emergency, manufacturing and approval of vaccines can be streamlined through an Emergency Use Authorization or EUA. An EUA does not affect vaccine safety, because it does not impact development, such as research, clinical studies and the studying of side effects and adverse reactions. Instead, it speeds up manufacturing and administrative processes.
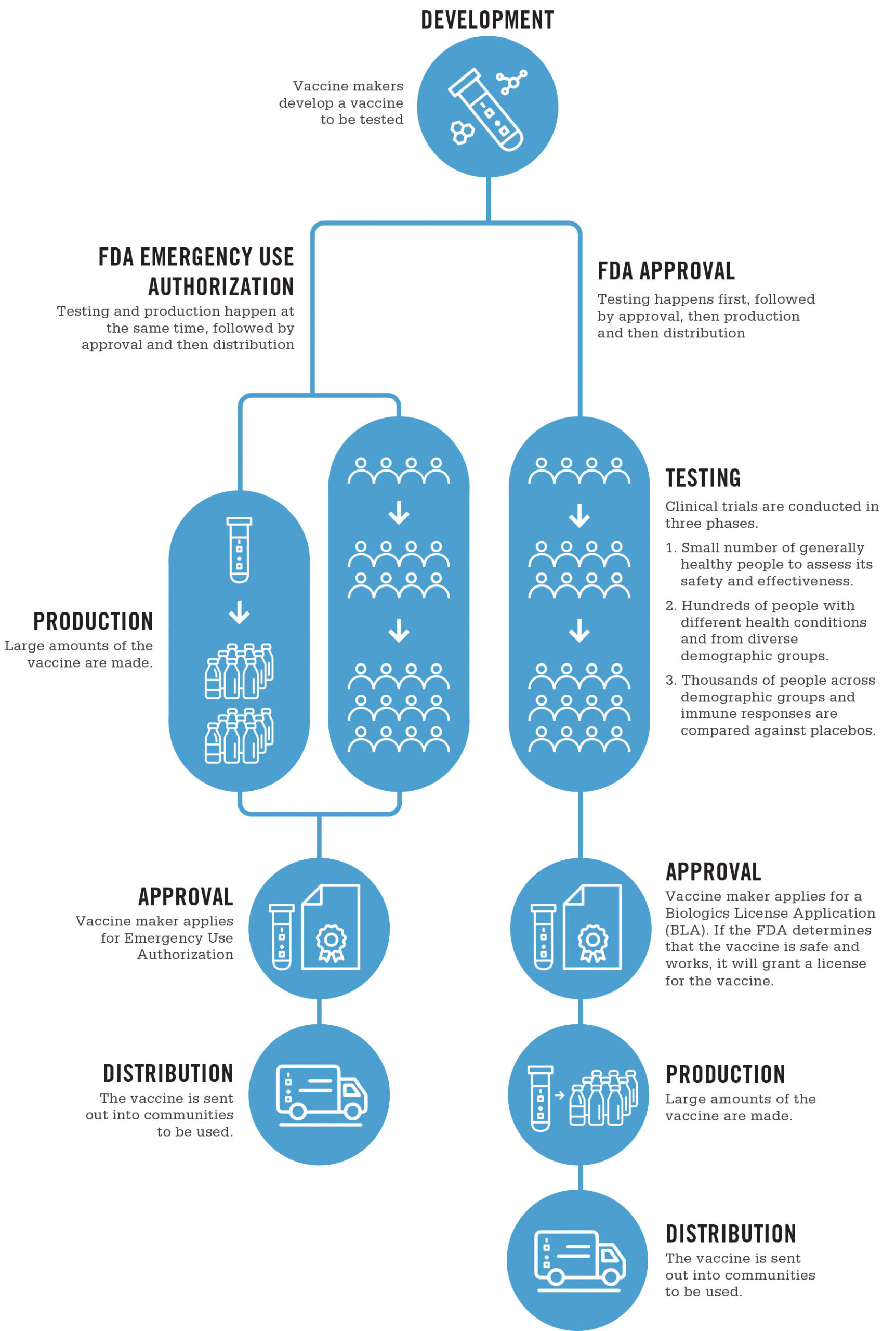
All vaccines follow the same testing processes, whether they are approved for emergency use or through a typical license.
Clinical trials are conducted in three phases.
- In Phase 1, the vaccine is given to a small number of generally healthy people to assess its safety and effectiveness.
- In Phase 2, the vaccine is given to hundreds of people with different health conditions and from diverse demographic groups.
- In Phase 3, the vaccine is administered to thousands of people across demographic groups and immune responses are compared against placebos, which are doses that don’t contain any of the vaccine and are used for testing purposes only.
Once Phase 3 trials are complete, the FDA reviews the data to determine whether the vaccine works and is safe. If so, the manufacturer files for approval.
This is where the process may change due to a public health emergency, such as the COVID-19 pandemic.
Usually, the manufacturer would apply for a Biologics License Application (BLA). If the FDA determines that the vaccine is safe, works and that manufacturing can be done safely and consistently, it will grant a license for the vaccine.
In a public health emergency, manufacturing may occur while vaccines are still in development, rather than after approval. These efforts happen simultaneously, and instead of filing for a BLA, the manufacturer files for Emergency Use Authorization.
If the benefits outweigh any possible risks of the vaccine and manufacturing quality can be ensured, the FDA will approve the vaccine for emergency use.
Emergency use authorizations are an important part in addressing public health emergencies and ending the COVID-19 pandemic.